「the clinical」の検索結果
88件:76~80件目を表示
もしかして
-
お知らせ | 千寿製薬株式会社
eMED”(PDF) January 17, 2025 Senju has Applied for Manufacturing and Marketing Approval in Japan for the novel Dry Eye Disease treatment, SJP-0132(PDF) November 26, 2024 Launch and Distribution of “MIE...
http://www.senju.co.jp/english/news/ -
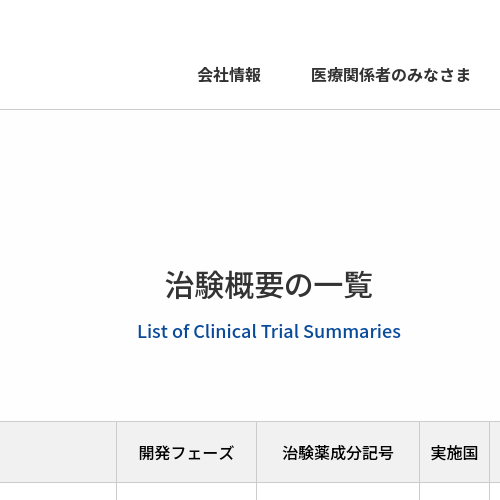
治験情報 | 研究開発活動 | 千寿製薬株式会社
治験概要の一覧List of Clinical Trial Summaries ※初回公表日の新しい順に掲載 対象疾患 開発フェーズ 治験薬成分記号 実施国 実施状況 臨床試験ID アレルギー性結膜炎 2 SJP-0049 日本 研究終了 jRCT2031250103 ドライアイ 3 SJP-0132 中国 研究終了 NCT06717152 健康成人 1 SJP-0170 日本 研究終了 jRCT...
https://www.senju.co.jp/rd/trial.html -
Ethical Products for Medical Professionals | Business Activities | About Senju | SENJU Pharmaceutical
The success of our premier line of ophthalmological drugs has launched our entry into other promising fields of medicine Senju Pharmaceutical greatly values the clinical experience of medical professi...
https://www.senju.co.jp/english/about/business/mp.html -

g approval in Japan as of December 22, 2025. Avarept® Ophthalmic suspension 0.3% represents a novel therapy for DED with Transient Receptor Potential cation channel subfamily V member 1 (TRPV1)*1 anta...
https://www.senju.co.jp/system/files/content_news/2025-12/ENG_20251222_0.pdf -

g approval in Japan as of December 22, 2025. Avarept® Ophthalmic suspension 0.3% represents a novel therapy for DED with Transient Receptor Potential cation channel subfamily V member 1 (TRPV1)*1 anta...
https://www.senju.co.jp/system/files/content_news/2025-12/ENG_20251222.pdf






